Abstract
Objective
This study aimed to evaluate the protective effect of igalan, a sesquiterpene lactone isolated from Inula helenium (L.), on inhibiting inflammation, regulating the epidermal differentiation gene expression, and reactive oxygen species scavenging in atopic dermatitis (AD)-like inflammatory keratinocytes.
Methods
HaCaT human keratinocytes were treated with igalan at indicated concentrations before being activated by a combination of TNF-α and IFN-γ or IL-4 representative for T-helper 1 and T-helper 2 cell cytokines, which are associated with AD pathogenesis.
Results
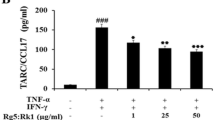
By inhibiting the NF-κB pathway as well as the STAT activation, igalan could downregulate several marker inflammatory genes in AD, such as TARC/CCL17, MDC/CCL22, and RANTES/CCL5. In contrast, igalan, acting as JAK inhibitor, could promote the mRNA expression levels of the genes FLG, LOR, KRT10, and DSC1, which encode for essential proteins responsible for keratinocyte differentiation, by inhibiting STAT3 signaling. Furthermore, igalan exerts its antioxidant effect through activating the Nrf2 pathway, triggering the expression of some enzymes that contribute to preventing intracellular ROS generation during inflammation.
Conclusion
These findings indicate that igalan, via suppressing JAK/STAT3 signaling, could impair the production of pro-inflammatory chemokines and enhance expression levels of several genes involved in keratinocyte differentiation in AD-like stimulated keratinocytes.






Similar content being viewed by others
References
Dainichi T, Hanakawa S, Kabashima K. Classification of inflammatory skin diseases: a proposal based on the disorders of the three-layered defense systems, barrier, innate immunity and acquired immunity. J Dermatol Sci. 2014;76(2):81–9. https://doi.org/10.1016/j.jdermsci.2014.08.010.
Furue M, Chiba T, Tsuji G, Ulzii D, Kido-Nakahara M, Nakahara T, Kadono T. Atopic dermatitis: immune deviation, barrier dysfunction, IgE autoreactivity and new therapies. Allergol Int. 2017;66(3):398–403. https://doi.org/10.1016/j.alit.2016.12.002.
Wong ITY, Tsuyuki RT, Cresswell-Melville A, Doiron P, Drucker AM. Guidelines for the management of atopic dermatitis (eczema) for pharmacists. Can Pharm J (Ott). 2017;150(5):285–97. https://doi.org/10.1177/1715163517710958.
Murota H, Takeuchi S, Sugaya M, Tanioka M, Onozuka D, Hagihara A, Saeki H, Imafuku S, Abe M, Shintani Y, Kaneko S, Masuda K, Hiragun T, Inomata N, Kitami Y, Tsunemi Y, Abe S, Kobayashi M, Morisky DE, Furue M, Katoh N. Characterization of socioeconomic status of Japanese patients with atopic dermatitis showing poor medical adherence and reasons for drug discontinuation. J Dermatol Sci. 2015;79(3):279–87. https://doi.org/10.1016/j.jdermsci.2015.05.010.
Hengge UR, Ruzicka T, Schwartz RA, Cork MJ. Adverse effects of topical glucocorticosteroids. J Am Acad Dermatol. 2006;54(1):1–15. https://doi.org/10.1016/j.jaad.2005.01.010.
Saaf AM, Tengvall-Linder M, Chang HY, Adler AS, Wahlgren CF, Scheynius A, Nordenskjold M, Bradley M. Global expression profiling in atopic eczema reveals reciprocal expression of inflammatory and lipid genes. PLoS ONE ONE. 2008. https://doi.org/10.1371/journal.pone.0004017.
Giridharan S, Srinivasan M. Mechanisms of NF-kappaB p65 and strategies for therapeutic manipulation. J Inflamm Res. 2018;11:407–19. https://doi.org/10.2147/JIR.S140188.
Bao L, Zhang H, Chan LS. The involvement of the JAK-STAT signaling pathway in chronic inflammatory skin disease atopic dermatitis. JAKSTAT. 2013. https://doi.org/10.4161/jkst.24137.
Klonowska J, Gleń J, Nowicki RJ, Trzeciak M. New cytokines in the pathogenesis of atopic dermatitis—new therapeutic targets. Int J Mol Sci. 2018;19:3086.
Omata N, Tsukahara H, Ito S, Ohshima Y, Yasutomi M, Yamada A, Jiang M, Hiraoka M, Nambu M, Deguchi Y, Mayumi M. Increased oxidative stress in childhood atopic dermatitis. Life Sci. 2001;69(2):223–8.
Nakai K, Yoneda K, Maeda R, Munehiro A, Fujita N, Yokoi I, Moriue J, Moriue T, Kosaka H, Kubota Y. Urinary biomarker of oxidative stress in patients with psoriasis vulgaris and atopic dermatitis. J Eur Acad Dermatol Venereol. 2009;23(12):1405–8.
Lee KM, Shin JM, Chun J, Song K, Nho CW, Kim YS. Igalan induces detoxifying enzymes mediated by the Nrf2 pathway in HepG2 cells. J Biochem Mol Toxicol. 2019. https://doi.org/10.1002/jbt.22297.
Moon KY, Hahn BS, Lee J, Kim YS. A cell-based assay system for monitoring NF-kappaB activity in human HaCat transfectant cells. Anal Biochem. 2001;292(1):17–211. https://doi.org/10.1006/abio.2001.5059.
Chun J, Choi RJ, Khan S, Lee DS, Kim YC, Nam YJ, Lee DU, Kim YS. Alantolactone suppresses inducible nitric oxide synthase and cyclooxygenase-2 expression by down-regulating NF-kappaB, MAPK and AP-1 via the MyD88 signaling pathway in LPS-activated RAW 264.7 cells. Int Immunopharmacol. 2012;14(4):375–83. https://doi.org/10.1016/j.intimp.2012.08.011.
O'Shea JJ, Plenge R. JAK and STAT signaling molecules in immunoregulation and immune-mediated disease. Immunity. 2012;36(4):542–50. https://doi.org/10.1016/j.immuni.2012.03.014.
Jeong SJ, Lim HS, Seo CS, Jin SE, Yoo SR, Lee N, Shin HK. Anti-inflammatory actions of herbal formula Gyejibokryeong-hwan regulated by inhibiting chemokine production and STAT1 activation in HaCaT cells. Biol Pharm Bull. 2015;38(3):425–34. https://doi.org/10.1248/bpb.b14-00660.
Horiuchi Y, Bae SJ, Katayama I. Overexpression of the suppressor of cytokine signalling 3 (SOCS3) in severe atopic dermatitis. Clin Exp Dermatol. 2006;31(1):100–4. https://doi.org/10.1111/j.1365-2230.2005.01979.x.
David M, Ford D, Bertoglio J, Maizel AL, Pierre J. Induction of the IL-13 receptor alpha2-chain by IL-4 and IL-13 in human keratinocytes: involvement of STAT6, ERK and p38 MAPK pathways. Oncogene. 2001;20(46):6660–8. https://doi.org/10.1038/sj.onc.1204629.
Sun Y, Lu Q, He L, Shu Y, Zhang S, Tan S, Tang L. Active fragment of Veronica ciliata Fisch. attenuates t-BHP-induced oxidative stress injury in HepG2 cells through antioxidant and antiapoptosis activities. Oxid Med Cell Longev. 2017. https://doi.org/10.1155/2017/4727151.
Morgan MJ, Liu Z-g. Crosstalk of reactive oxygen species and NF-κB signaling. Cell Res. 2010;21:103. https://doi.org/10.1038/cr.2010.178.
Leung DYM, Boguniewicz M, Howell MD, Nomura I, Hamid QA. New insights into atopic dermatitis. J Clin Investig. 2004;113(5):651–7. https://doi.org/10.1172/JCI21060.
Danso MO, van Drongelen V, Mulder A, van Esch J, Scott H, van Smeden J, El Ghalbzouri A, Bouwstra JA. TNF-α and Th2 cytokines induce atopic dermatitis-like features on epidermal differentiation proteins and stratum corneum lipids in human skin equivalents. J Investig Dermatol. 2014;134(7):1941–50. https://doi.org/10.1038/jid.2014.83.
De Vuyst E, Salmon M, Evrard C, de Rouvroit CL, Poumay Y. Atopic dermatitis studies through in vitro models. Front Med. 2017. https://doi.org/10.3389/fmed.2017.00119.
Lim HS, Yo SR, Lee MY, Seo CS, Shin HK, Jeong SJ. Potential inhibitory effects of the traditional herbal prescription Hyangso-san against skin inflammation via inhibition of chemokine production and inactivation of STAT1 in HaCaT keratinocytes. Mol Med Rep. 2018;17(2):2515–22. https://doi.org/10.3892/mmr.2017.8172.
Gluck J, Rogala B. Chemokine RANTES in atopic dermatitis. Arch Immunol Ther Exp (Warsz). 1999;47(6):367–72.
Shimada Y, Takehara K, Sato S. Both Th2 and Th1 chemokines (TARC/CCL17, MDC/CCL22, and Mig/CXCL9) are elevated in sera from patients with atopic dermatitis. J Dermatol Sci. 2004;34(3):201–8. https://doi.org/10.1016/j.jdermsci.2004.01.001.
Thijs JL, de Bruin-Weller MS, Hijnen D. Current and future biomarkers in atopic dermatitis. Immunol Allergy Clin North Am. 2017;37(1):51–61. https://doi.org/10.1016/j.iac.2016.08.008.
Leyva-Castillo JM, Hener P, Jiang H, Li M. TSLP produced by keratinocytes promotes allergen sensitization through skin and thereby triggers atopic march in mice. J Invest Dermatol. 2013;133(1):154–63. https://doi.org/10.1038/jid.2012.239.
Seki Y, Inoue H, Nagata N, Hayashi K, Fukuyama S, Matsumoto K, Komine O, Hamano S, Himeno K, Inagaki-Ohara K, Cacalano N, O'Garra A, Oshida T, Saito H, Johnston JA, Yoshimura A, Kubo M. SOCS-3 regulates onset and maintenance of T(H)2-mediated allergic responses. Nat Med. 2003;9(8):1047–54. https://doi.org/10.1038/nm896.
Goren I, Linke A, Muller E, Pfeilschifter J, Frank S. The suppressor of cytokine signaling-3 is upregulated in impaired skin repair: implications for keratinocyte proliferation. J Invest Dermatol. 2006;126(2):477–85. https://doi.org/10.1038/sj.jid.5700063.
Candi E, Schmidt R, Melino G. The cornified envelope: a model of cell death in the skin. Nat Rev Mol Cell Biol. 2005;6(4):328–40. https://doi.org/10.1038/nrm1619.
Omori-Miyake M, Yamashita M, Tsunemi Y, Kawashima M, Yagi J. In vitro assessment of IL-4- or IL-13-mediated changes in the structural components of keratinocytes in mice and humans. J Invest Dermatol. 2014;134(5):1342–50. https://doi.org/10.1038/jid.2013.503.
Wang Q, Gao S, Wu G-Z, Yang N, Zu X-P, Li W-C, Xie N, Zhang R-R, Li C-W, Hu Z-L, Zhang W-D. Total sesquiterpene lactones isolated from Inula helenium L. attenuates 2,4-dinitrochlorobenzene-induced atopic dermatitis-like skin lesions in mice. Phytomedicine. 2018;46:78–84. https://doi.org/10.1016/j.phymed.2018.04.036.
Acknowledgements
This study was supported by grants (NRF-2017R1A2B4009301) from the National Research Foundation of Korea and Korea Institute of Science and Technology (KIST370C-20190043). We appreciate the technical support from Ms. Ji Yoon Lee, the College of Pharmacy, Seoul National University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Responsible Editor: John Di Battista.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dao, T.T.P., Song, K., Kim, J.Y. et al. Igalan from Inula helenium (L.) suppresses the atopic dermatitis-like response in stimulated HaCaT keratinocytes via JAK/STAT3 signaling. Inflamm. Res. 69, 309–319 (2020). https://doi.org/10.1007/s00011-020-01322-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-020-01322-4